KYPHOPLASTY PROCEDURE
HOME
>
SURGICAL PROCEDURES
>
SPINE SURGERY PROCEDURES
>
KYPHOPLASTY PROCEDURE
A vertebral compression fracture (VCF) occurs when the vertebral body fractures and collapses due to osteoporotic conditions, tumours (e.g. myeloma) or trauma.
The principle of kyphoplasty is to restore vertebral body anatomy gently and progressively by inflating balloons and then reinforcing the anterior column of the vertebra with cement. The balloons create a cavity within the vertebral body, compressing the cancellous bone and thus limiting the risk of cement leakage from the vertebral body.
The balloon catheter is then deflated, and the cavity created is now filled with bone cement, avoiding any leakage.
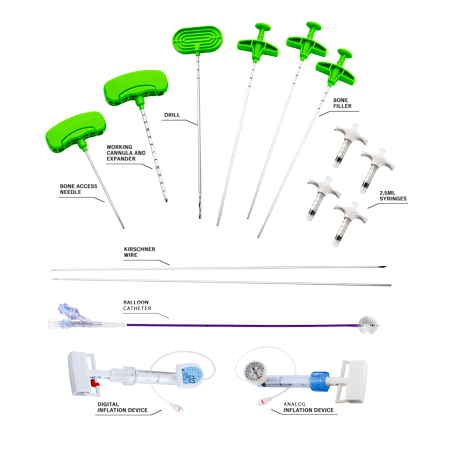
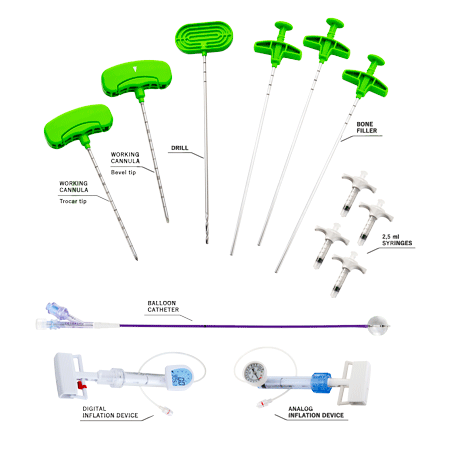
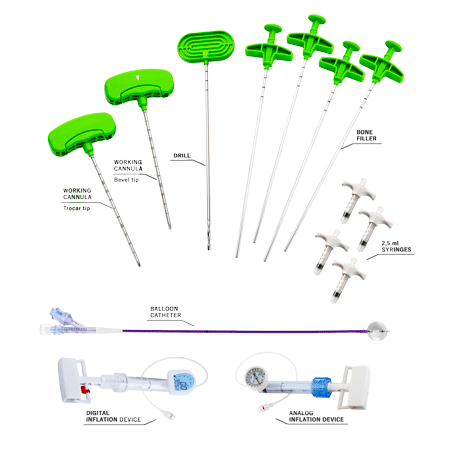
RENOVA SPINE™ is a minimally invasive system for kyphoplasty procedure to treat fractures, restore vertebral body height and reduce back pain. RENOVA SPINE™ system is designed to reduce fractures and/or to creation cavities in the spongy bone of the spine and is intended to be used in combination with a legally marketed bone void filler indicated for use in vertebroplasty or vertebral filling procedures.
There are two access options, either using a working cannula with a trocar/bevel tip or using a guidewire. The working cannula allows to carry out a direct percutaneous vertebral access with fewer surgical steps. This device is part of the RENOVA SPINE™ 11G Kyphoplasty kit and RENOVA SPINE™ 13G Kyphoplasty kit.
The guidewire is used to guide the introduction of the following instruments and is sold within the RENOVA SPINE™ 8G Kyphoplasty kit.
INDICATIONS:
- Painful, vertebral compression fractures.
- Treatment of osteolytic lesions within the vertebral body.
KYPHOPLASTY BENEFITS
BENEFITS
Minimally invasive solution
Patient recovers faster.
Percutaneous
Less invasive and less traumatic treatment, reduced muscle and soft tissue damage, leading to less post-operative pain.
Cavity creation
Minimized risk of cement leakage, as compressed cancellous bone acts as natural barrier to cement. Cement of higher viscosity can be injected.
Height restoration
Restoration of vertebral body height, re-alignment and restoration of physiological alignment of the spine.
Height conservation
Physiological alignment of the spine according to the patient’s natural anatomy decreases the risk of adjacent level fractures and co-morbidities.
More flexibility. The implantation of a metallic implant is technically more demanding for exact correct positioning.
Reduction in morbidity
The benefit has been demonstrated in terms of alleviation of pain and fast functional improvement.
SURGICAL PROCEDURE
It is possible to perform a transpedicular or extrapedicular access, depending on the anatomy of the vertebral body to be treated.
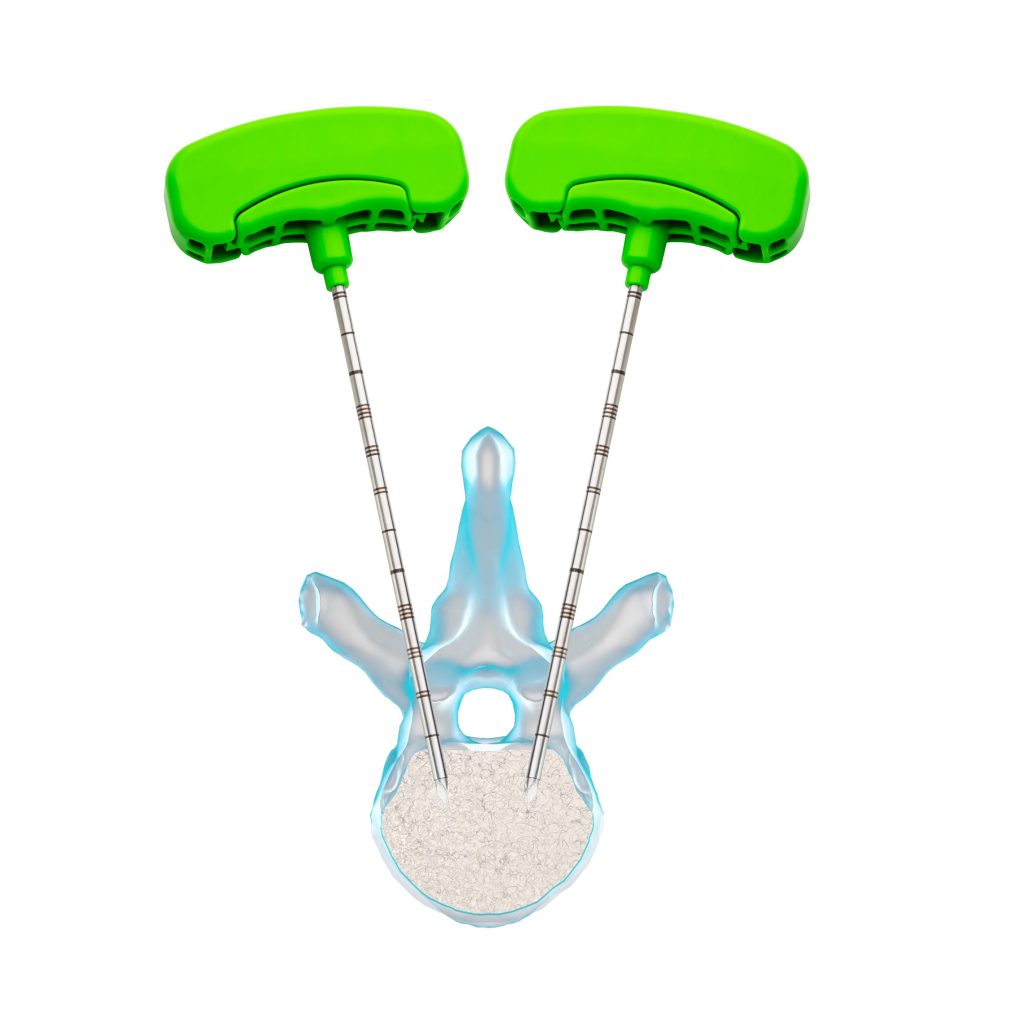
1- Access creation: under fluoroscopy, create an access channel, either by means of a working cannula with a trocar/bevel tip or by means of a guidewire. If necessary, tap the plastic handle of the working cannula with a hammer to let the instrument advance.
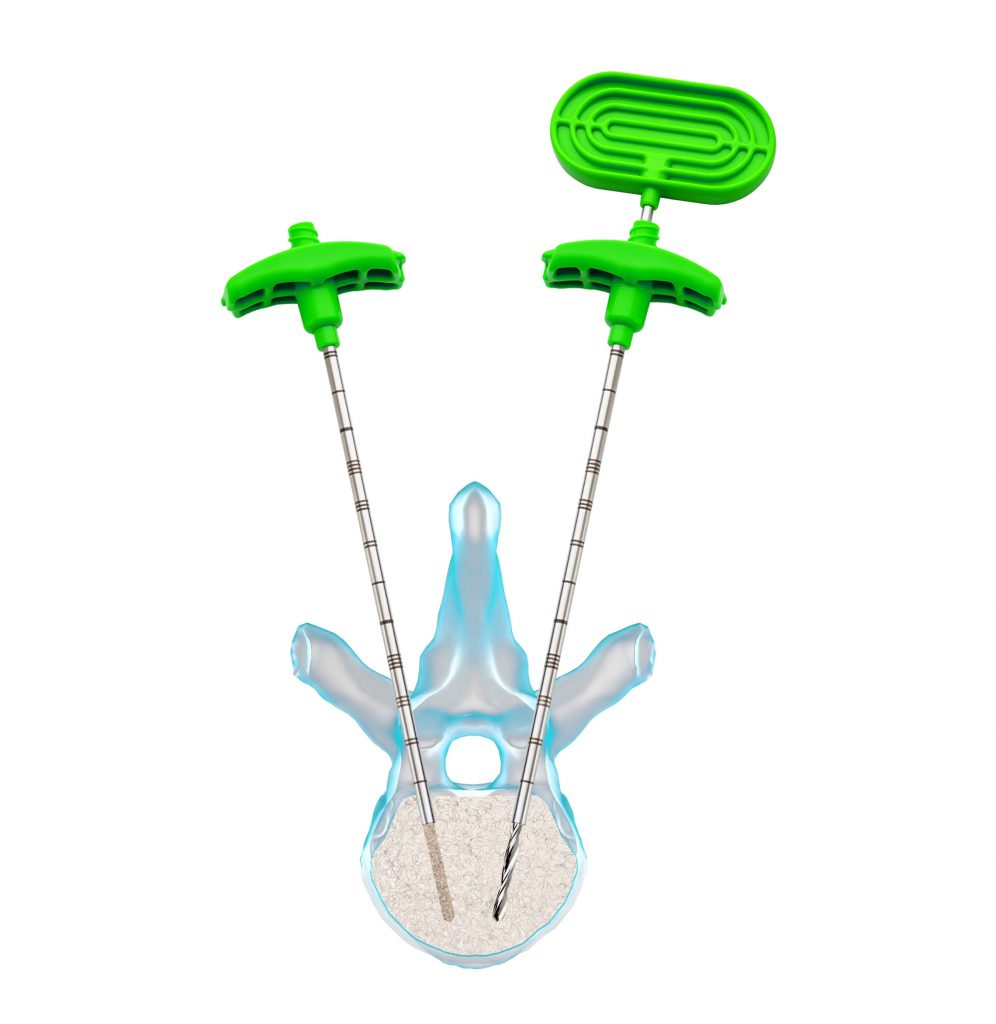
2 – Prepare the cavity: remove the internal stylet of the working cannula, leaving only the cannula inside the vertebral body. Insert the drill into the cannula and then into the vertebral body. Rotate 180° clockwise and counterclockwise to create the cavity.

3- Define the balloon size: Renova Spine Kyphoplasty Drill has depth marks or notches that serve as a reference.
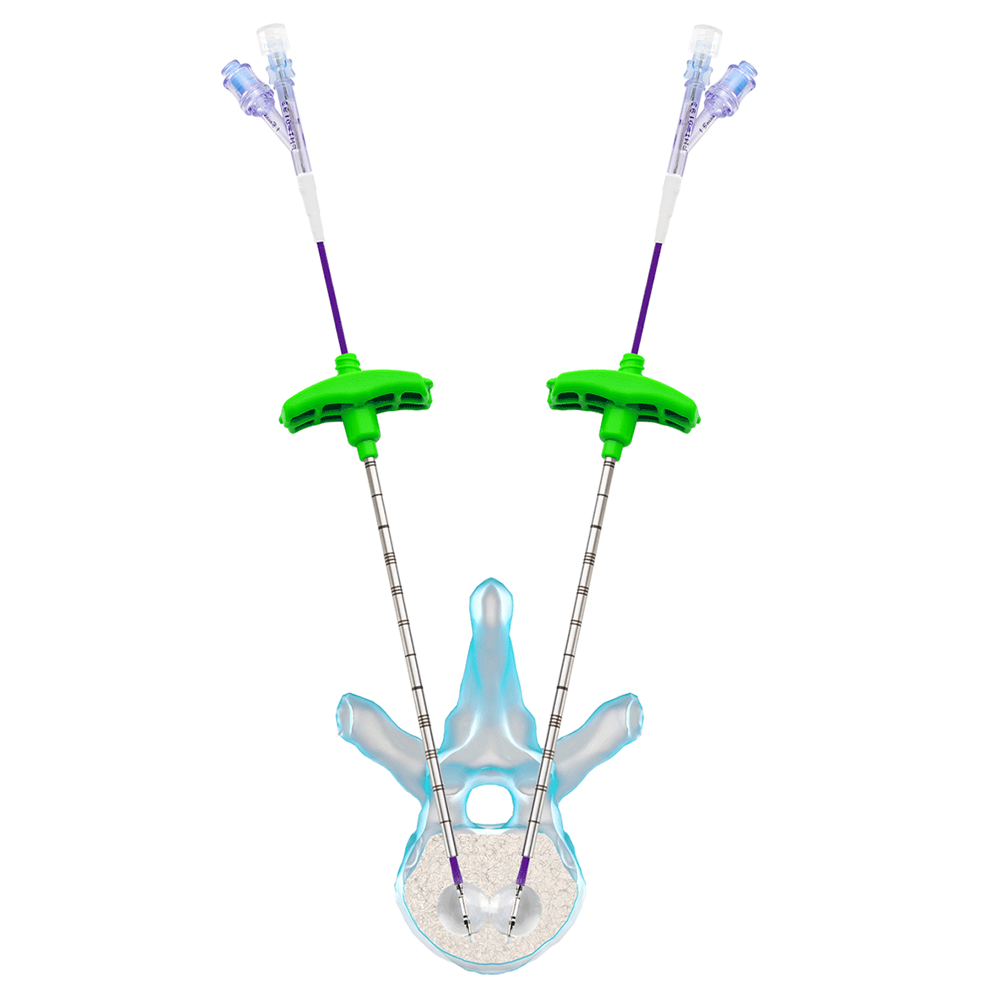
4 – Prepare the inflation device: load the inflation device with a contrast medium. Insert the balloon catheter, under fluoroscopic guidance, through the working cannula and reach the point where you want to expand it. Connect the inflation system to the side door of the catheter.
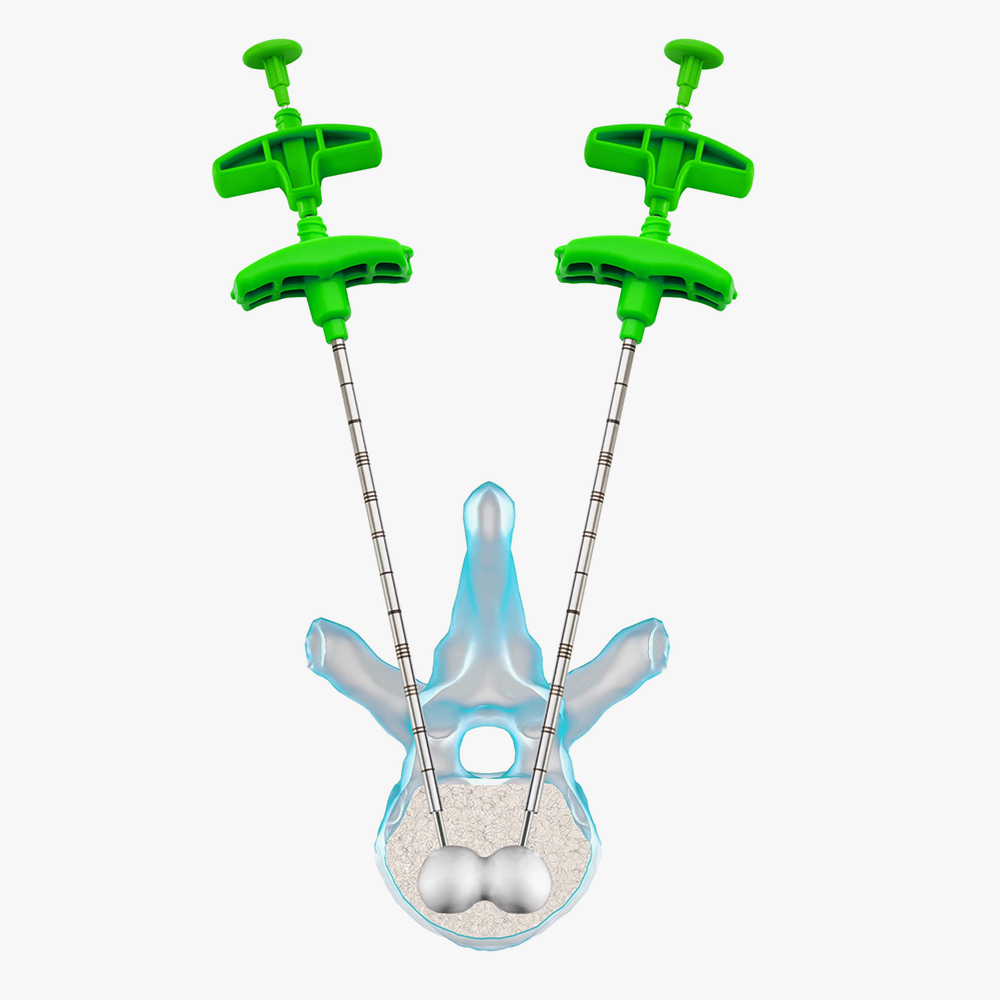
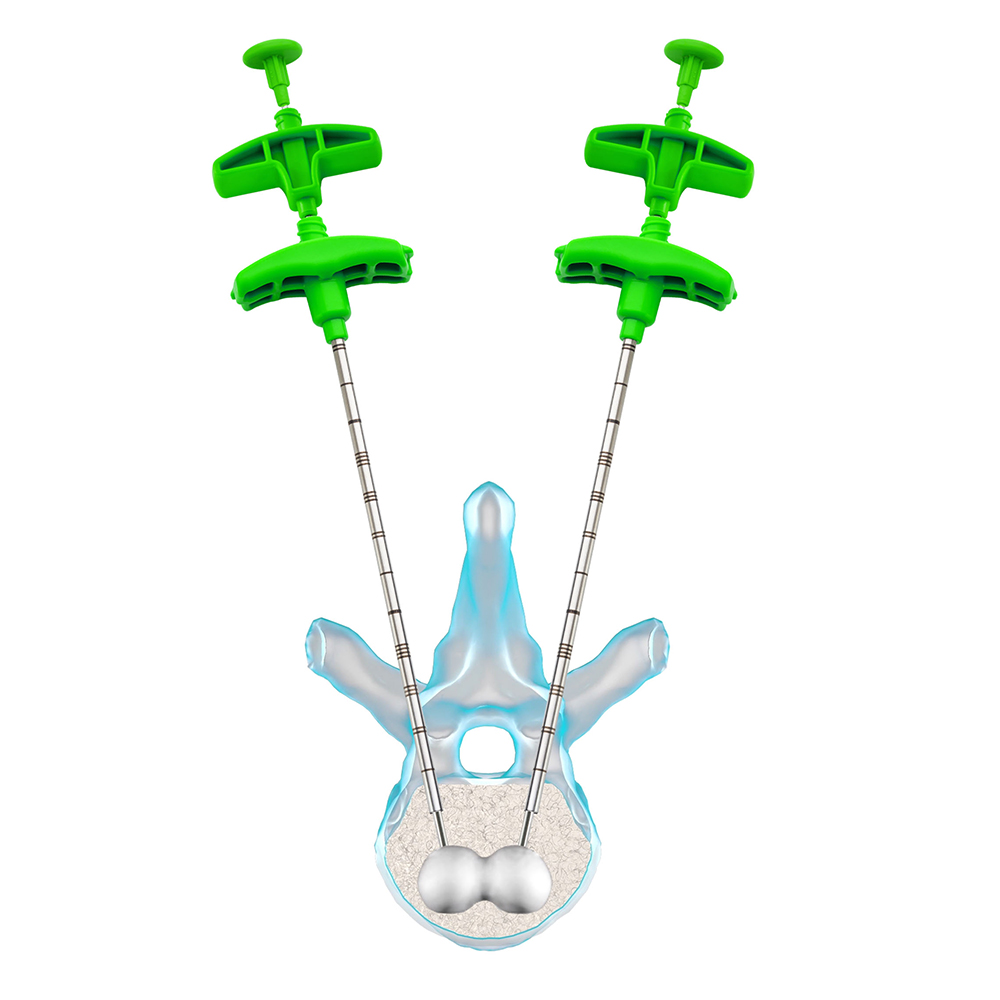
5 – Create the cavity: inflate the balloon under fluoroscopy till the desired volume is reached.
6 – Fill the cavity with PMMA cement: load the cement injection cannula with cement and insert them one at a time into the working cannula and with the plunger proceed to the controlled injection of cement into the vertebral body.
RELATED PRODUCT
Discover more about RENOVA SPINE™ KYPHOPLASTY KIT:
VIDEO TUTORIAL
Fill out the form to request the complete RENOVA SPINE™ KYPHOPLASTY Surgical technique.