DISKOM™
Percutaneous discectomy system
- Minimally invasive percutaneous access for faster recovery;
- No thermal damage to nerve roots and surrounding tissues;
- Disc annulus integrity preservation;
- Rapid patient rehabilitation & fast discharge;
- Procedure time: 10-15 minutes;
- No capital investment or extra equipment required;
- Only local anaesthesia is necessary;
- Potentially up to 2 cc of disc material collected;
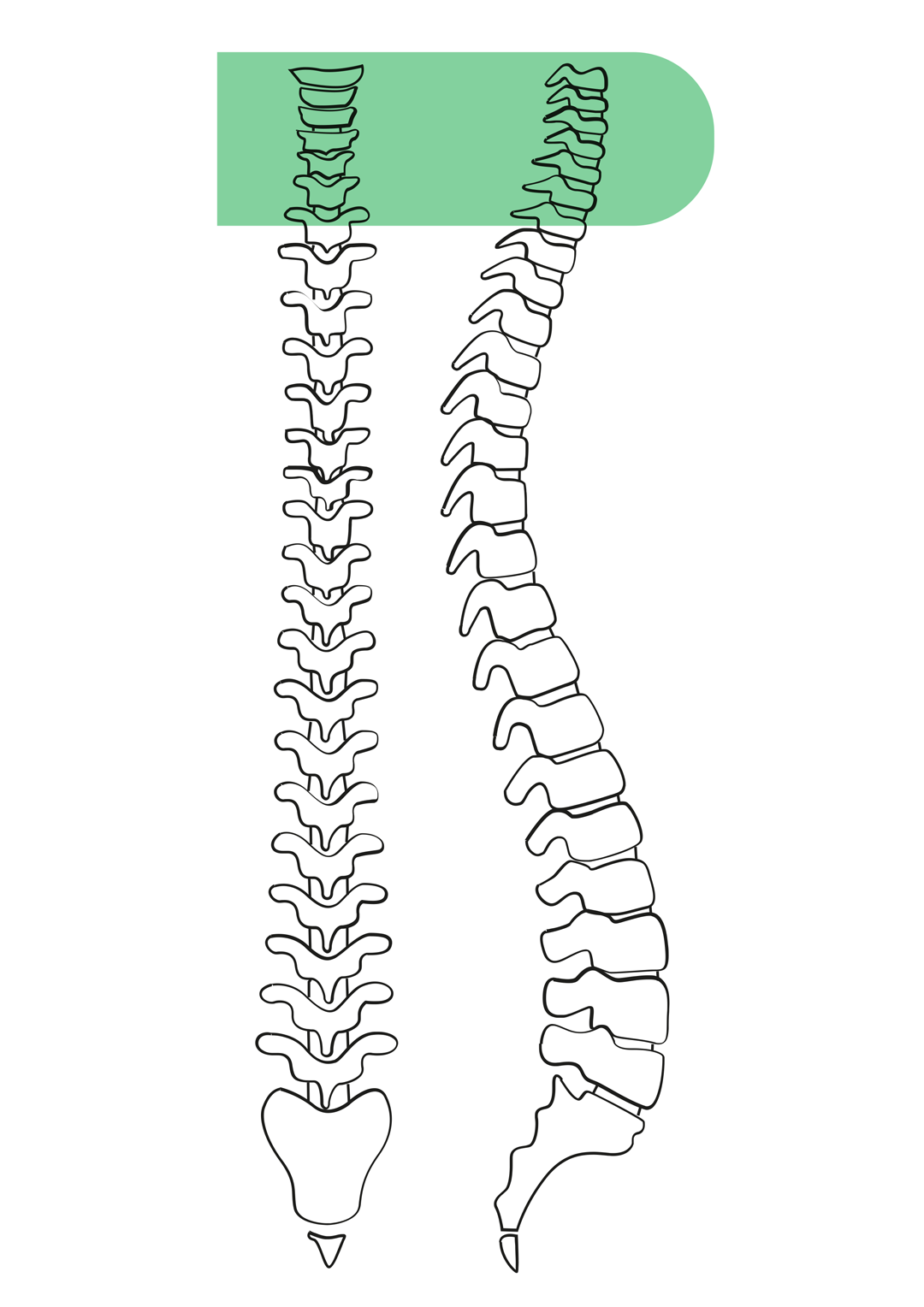
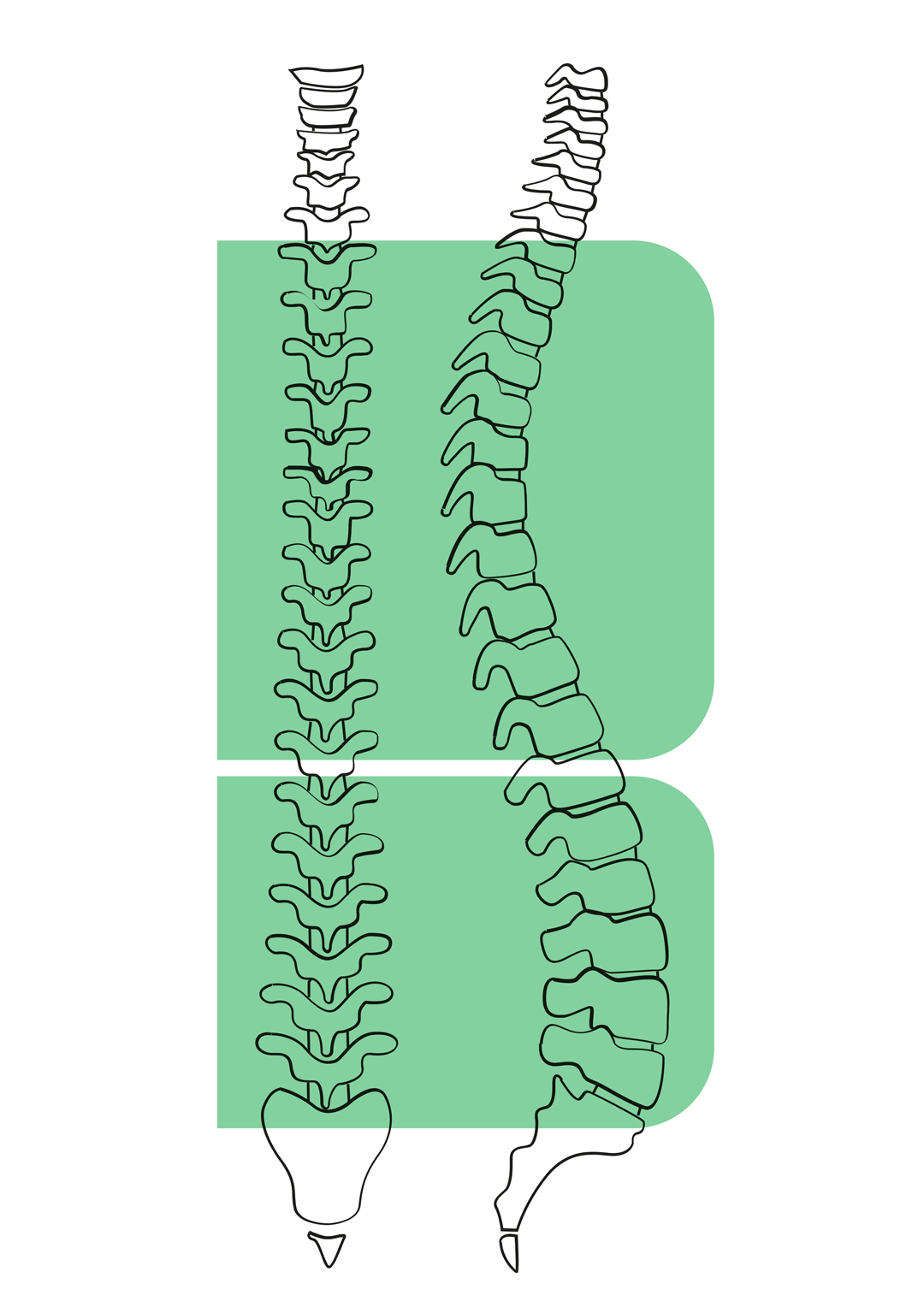
- Percutaneous discectomy system available for thoracolumbar and cervical spine;
- Single-use device with biopsic procedure possibility.
A hernia is a common disorder that occurs when the intervertebral disc’s nucleus pulposus becomes dislodged from its natural position inside the annulus fibrosus. Most annulus fibrosus lesions are the result of repeated microtraumas or a major trauma that degenerates to allow the nucleus pulposus to bulge and compress a nerve or its surrounding tissues, causing pain in the back and the legs. When conservative treatment fails, and symptoms persist or worsen, surgical treatment is considered.
Using the DISKOM™ percutaneous discectomy system, spine specialists can reduce pressure on the nerve root by removing the disc nucleus in a minimally invasive percutaneous procedure.
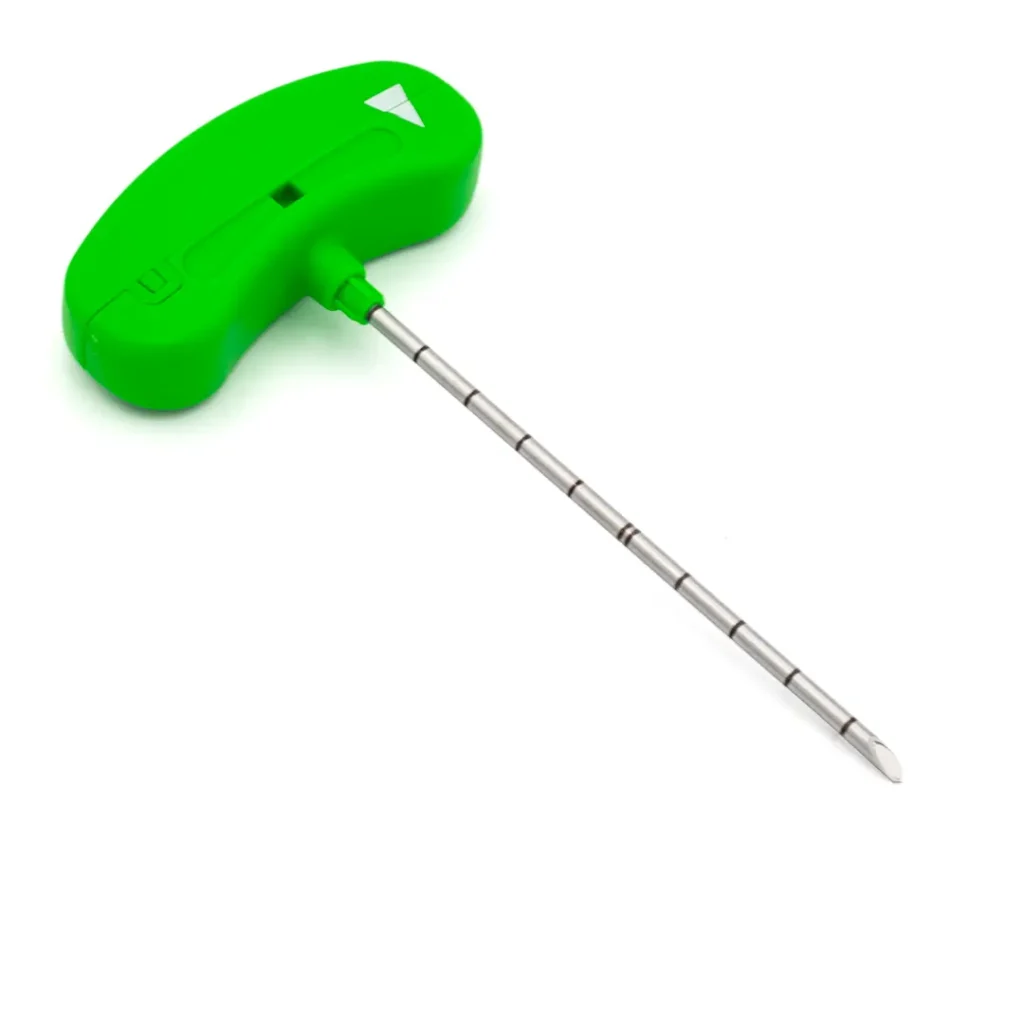
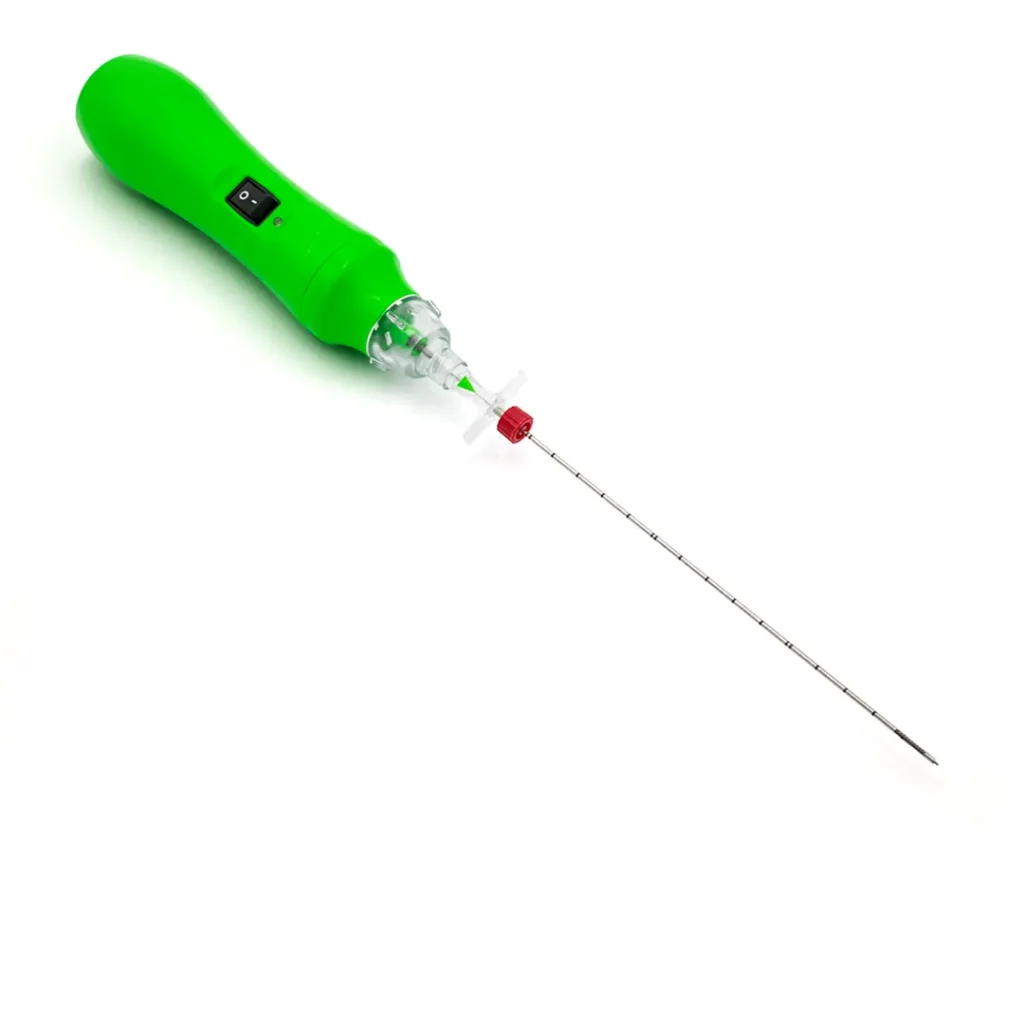
DISKOM™ is made up of two elements: an access needle with both distal and side openings, and a titanium cochlear tip for the mechanical removal of the nucleus pulposus. The major advantage of this device is that the cochlea is tightly connected, making detachment almost impossible; this is a unique feature of this percutaneous discectomy system.
It is a mechanical removal procedure. It does not make use of either radiofrequency or laser. The wide side opening enables the removal of potentially up to 2cc of disc material. The basic principle uses an Archimedes’ screw or cochlea.
SURGICAL TECHNIQUE (thoracolumbar percutaneous discectomy system)
- Under fluoroscopy, introduce the access needle with its stylet into the disc.
- Remove the stylet and introduce the probe into the cannula.
- Lock the probe to the access needle through the Luer-lock connection.
- Switch the probe on.
- Carry out a continuum movement in an anteroposterior direction for 2/3 minutes and simultaneously proceed with a rotary movement.
- Disc material is then removed and collected along the probe stylet or into the collection chamber.
- After switching off, remove the device.
SURGICAL TECHNIQUE (cervical percutaneous discectomy system)
- Under fluoroscopic guidance, identify the target disc and the safe anterior access corridor, between the carotid sheath laterally and the trachea–oesophageal plexus medially.
- After local anaesthesia, introduce the access needle with its stylet through the anterior approach, advancing carefully into the disc space under continuous lateral and anteroposterior fluoroscopic control.
- Remove the stylet and introduce the probe into the cannula.
- Lock the probe to the access needle through the Luer-lock connection.
- Switch the probe on.
- Perform a gentle anteroposterior movement for 1–2 minutes, combined with slow rotary movements, while maintaining stable needle positioning.
- Disc material is then removed and collected along the probe stylet or into the collection chamber.
- After switching off, remove the device.
| PROCEDURE | SIZES | LENGTH | CODES |
| Cervical | 19G | 8 cm | DKR1908CDFL10 |
| Thoracic / Lumbar | 17G | 16 cm | DKR1716CDFL10 |